You still have a little time.
On 30 November 2026, 365FarmNet will be discontinued.
What does this mean for you?
- You can start your switch to CLAAS connect now.
- Your contacts will remain the same.
- Your data can be migrated.
- The basic license is free for 365FarmNet customers for one year.
- Many functions are available immediately.
- Additional functions will be added continuously.

This is how the switch works
01
Get in touch
Your contact at your CLAAS dealer will be happy to help you.
Simply use our contact form.
02
Sign the changeover contract
You will receive CLAAS connect free of charge for one year. After that, the contract will renew automatically.
03
Technical migration
You provide your login details to your contact person and arrange an appointment for the migration.
04
Get access
You get started with your new CLAAS connect account.

What happens to my data?
To help you continue working smoothly, we support you with transferring your data. Together with your contact person, you can clarify what is transferred and how the process will work in detail.
- Together, we review your current situation (farm, machines, users).
- We agree on which data will be transferred.
- We carry out the data transfer together at the agreed appointment.
CLAAS connect at a glance
Easily manage your farm digitally
Enjoy automated documentation with machine and yield data
Manage mixed fleets seamlessly
Keep maintenance and service under control
Create application maps and reference tracks
Only pay for what you need with modular features
CLAAS connect at a glance
Easily manage your farm digitally
Enjoy automated documentation with machine and yield data
Manage mixed fleets seamlessly
Keep maintenance and service under control
Create application maps and reference tracks
Only pay for what you need with modular features
FAQ
A change always raises questions — that’s completely normal.
In this FAQ, we answer the most important questions about migrating from 365FarmNet to CLAAS connect. You can find more information about features and how to use CLAAS connect in the detailed FAQ on the CLAAS connect website.
With us, smart farming is part of everyday life.
Digital solutions for farm management, fleet management and precision farming: Get to know our farm management applications in the new CLAAS connect!
Your partner for farm management
365FarmNet offers comprehensive solutions for farm management: Since October 2024, our innovative applications are available in the new CLAAS connect across more than 30 countries worldwide — covering everything from operations management to fleet management and precision farming.
If you are already using 365FarmNet, you can continue to work as usual. Our customer service team will, of course, still be at your disposal. Please note that new registrations for 365FarmNet are no longer possible.
Join us in the future of connected farm management with CLAAS connect and benefit from innovative technologies for your operation.
365FarmNet in CLAAS connect
Our farm management solutions have excelled in practice, assisting our clients daily in their tasks. Drawing on our years of experience in agricultural software development and valuable practical feedback, we have co-developed the new CLAAS connect.
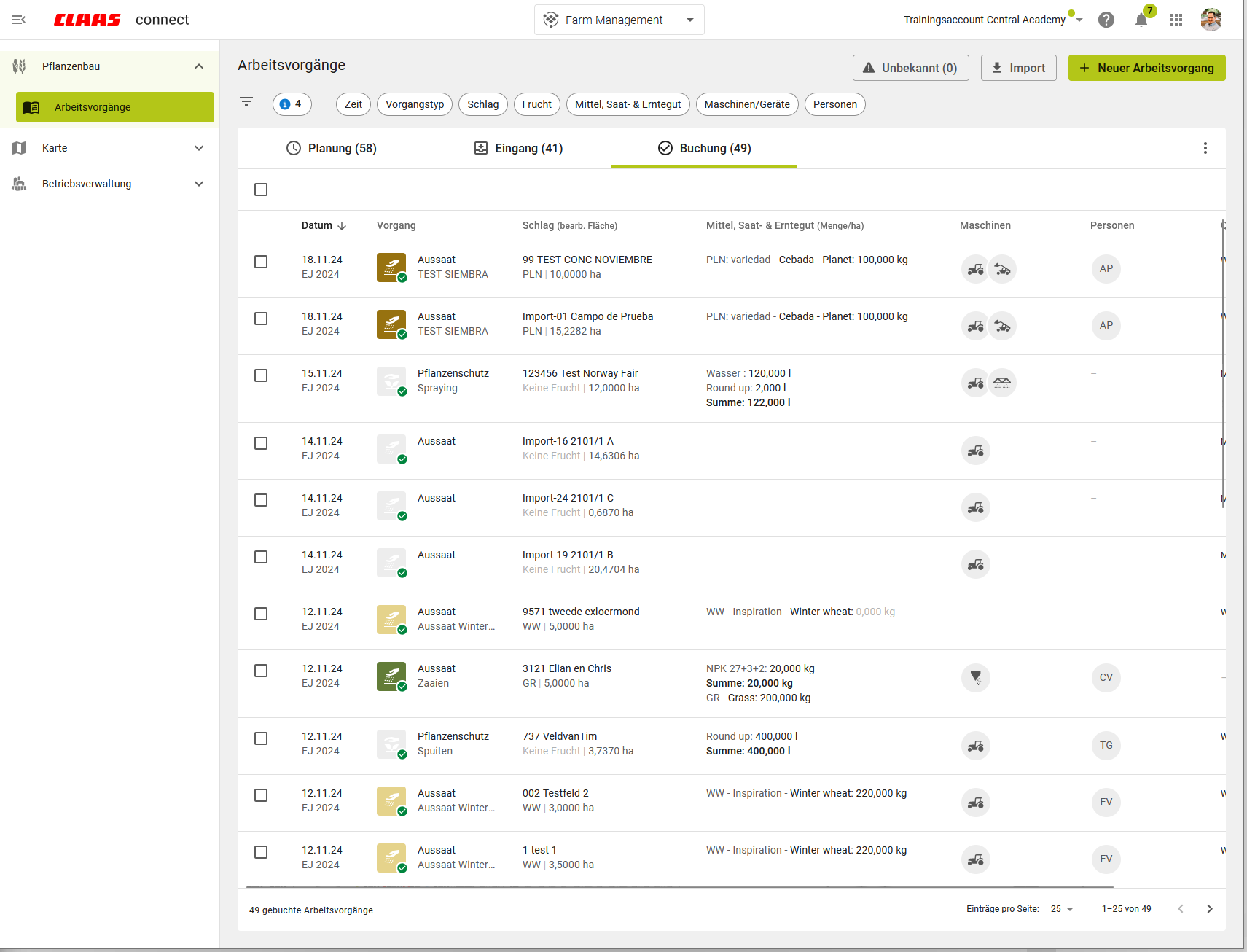
Task management: Proven solution redesigned
In the tradition of preserving and developing good things, we have designed task management in the new CLAAS connect.
Discover the advantages:
- More information in the task overview
- Improved filter functions
- New dialogue for integrating unknown master data
- New: Direct jump from the task to the geocenter for further processing of field data
- New: View additional counter values for all imported data in your files
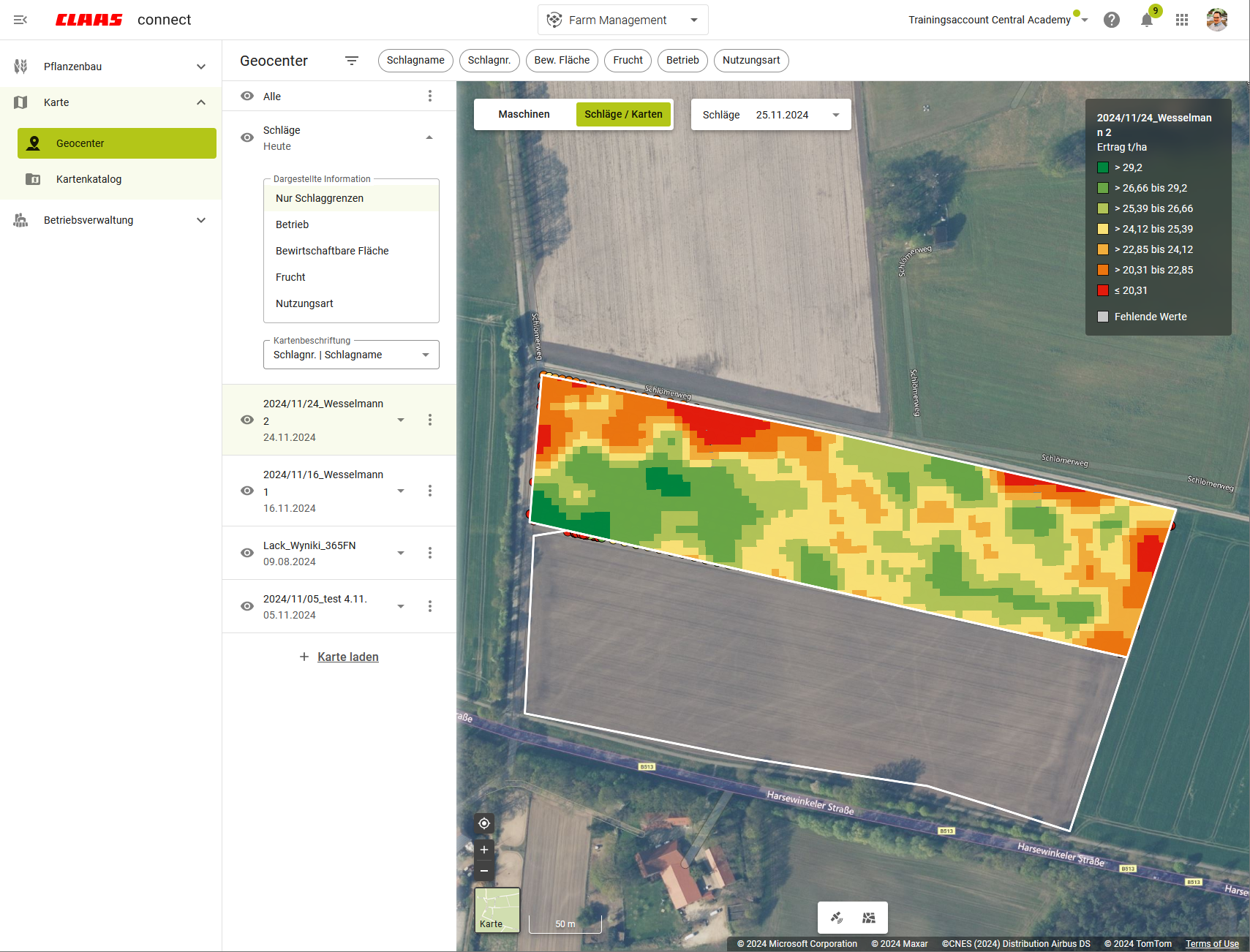
Precision Farming with the Geocenter
With the new CLAAS connect, agricultural businesses in over 30 countries benefit from our updated and expanded precision farming applications. CLAAS connect provides a central “toolbox” through the Geocenter for everything related to:
- Area and satellite data,
- Yield maps,
- Soil samples,
- Machine positions,
- Creating potential and application maps.
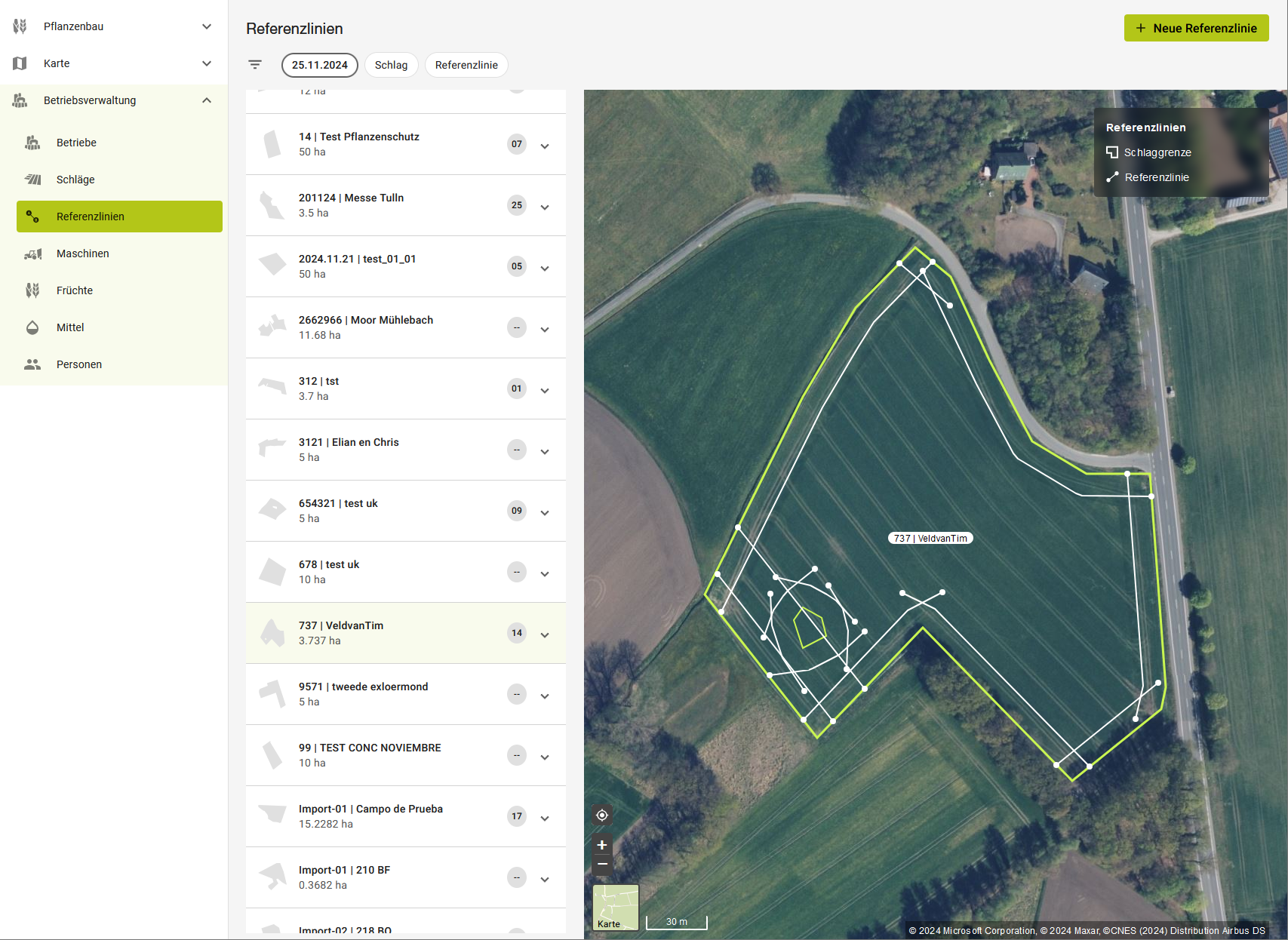
From the office to the equipment: Route planning and easy data transfer.
The new CLAAS connect takes route management and planning to a new level. All functionalities are now combined in one seamless application. Your planned routes are automatically integrated into the planned tasks and can be exported directly to your CEMIS 1200 if required.
Route management is available in the Farm Management section of the new CLAAS connect:
- Overview of all reference lines with and without field reference
- New: Quick view of the assigned machine, working width and turning radius
- Editing and duplication of reference lines
- Renaming of reference lines
- Assignment to a field
- Exporting and deleting a reference line
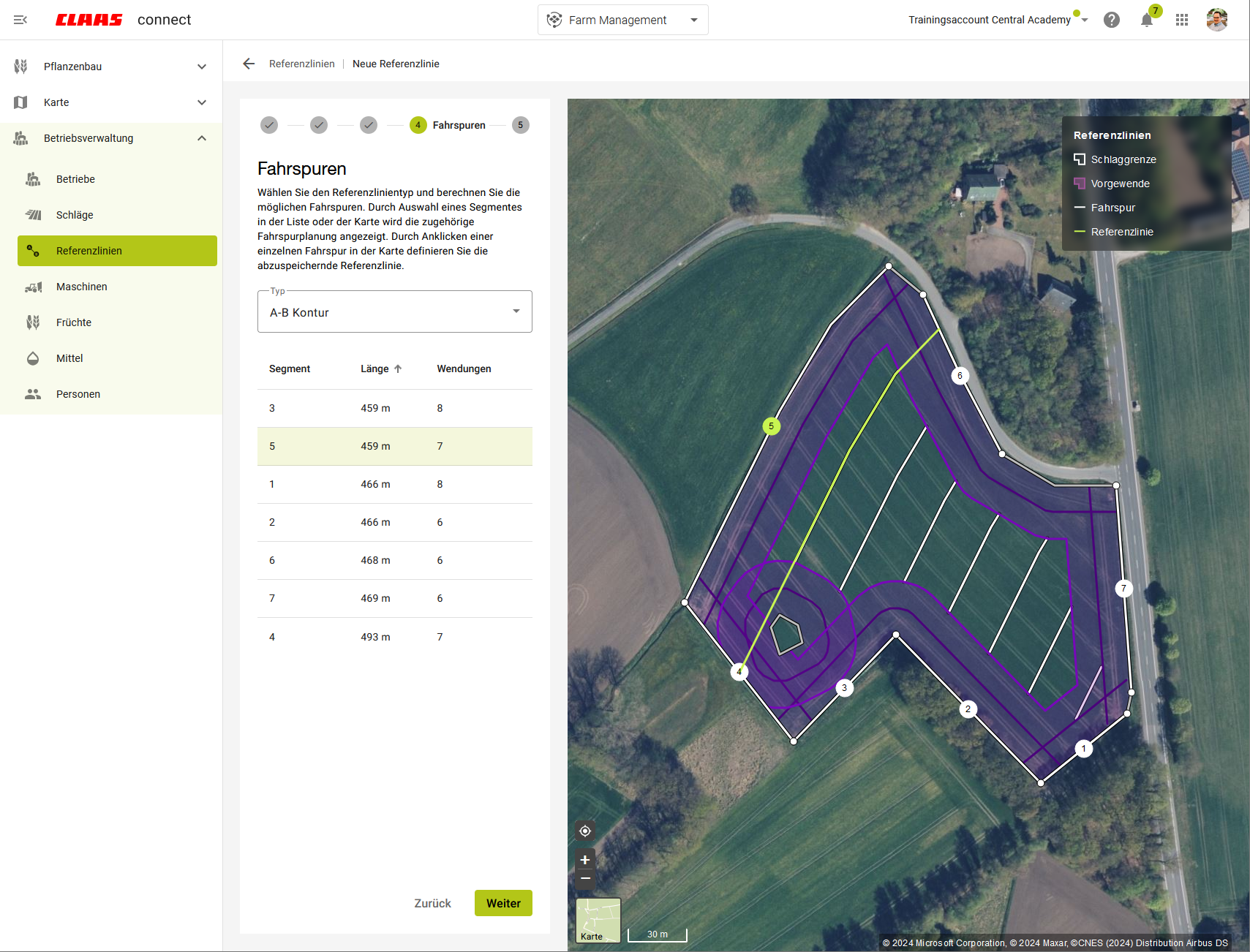
To facilitate seamless usage of reference lines, the creation is embedded directly within the reference line management:
- Selecting the machine performing the task for direct assignment when sending the order to a machine
- Considering the working width and turning radius
- Including headland lines in the planning
- Adjusting lines to the right and left
- Rotating lines at an angle to the right and left
Many new features support you every day.
Experience seamless integration from task planning to application maps, or from the map catalog to task execution. The continuous functions and high performance of the new CLAAS connect make managing and processing your data even easier now.

Discover the new CLAAS connect – Farm Management App – your comprehensive tool for all farm management functions, now conveniently combined in one place.
New features await you:
- Work progress display for CLAAS harvesting machines to estimate processing time
- Task management within the app towards CEMIS 1200
- Create markers on the map and synchronise them with all app users, e.g. field entrances or gully covers
- Send documentation as a PDF via email
- Processing of machine orders.